Carbetocin versus oxytocin for the prevention of postpartum haemorrhage following caesarean section: the results of a double-blind randomised trial
G Attilakos,a D Psaroudakis,a J Ash,a R Buchanan,a C Winter,a F Donald,a LP Hunt,b T Draycotta
a Directorate of Women’s and Children’s Health, Southmead Hospital, North Bristol NHS Trust, Bristol, UK b Department of Clinical Sciences at South Bristol, Institute of Child Life and Health, University of Bristol, Bristol, UK
Correspondence: Dr G Attilakos, Fetal Medicine Unit, St Michael’s Hospital, University Hospitals Bristol Foundation NHS Trust, Bristol BS2 8EG, UK. Emails georgeattilakos@doctors.org.uk, georgeattilakos@hotmail.com
Accepted 29 March 2010. Published Online 19 May 2010.
Objective To compare the effectiveness of carbetocin and oxytocin when they are administered after caesarean section for prevention of postpartum haemorrhage (PPH).
Study design Double-blind randomised single centre study (1:1 ratio).
Setting Teaching hospital in Bristol, UK with 6000 deliveries per annum.
Population Women at term undergoing elective or emergency caesarean section under regional anaesthesia, excluding women with placenta praevia, multiple gestation and placental abruption.
Methods Women were randomised to receive either carbetocin 100 lg or oxytocin 5 IU intravenously after the delivery of the baby. Perioperative care was otherwise normal and use of additional oxytocics was at the discretion of the operating obstetrician. Analysis was by intention to treat.
Primary outcome measure The proportion of women in each arm of the trial that needed additional pharmacological oxytocic interventions.
Results Significantly more women needed additional oxytocics in the oxytocin group (45.5% versus 33.5%, Relative risk 0.74, 95% CI 0.57–0.95). The majority of women had oxytocin infusions. There were no significant differences in the secondary outcomes, including major PPH, blood transfusions and fall in haemoglobin.
Conclusions Carbetocin is associated with a reduced use of additional oxytocics. It is unclear whether this may reduce rates of PPH and blood transfusions.
Keywords Caesarean section, carbetocin, oxytocin, postpartum haemorrhage.
Introduction
The administration of oxytocics after the delivery of the neonate reduces the likelihood of PPH5 and 5 IU oxytocin by slow intravenous injection is currently recommended in the UK for all caesarean sections.6 However, the use of additional oxytocic medication is common,7 to arrest bleeding, or prophylactically if there are risk factors for PPH. An audit in our hospital revealed that an additional 4-hour oxytocin infusion was used in more than 20% of caesarean sections.
Carbetocin is a synthetic analogue of human oxytocin with structural modifications that increase its half life thereby prolonging its pharmacological effects.8 Two dou- ble-blind randomised trials compared 100 lg carbetocin (the licensed dose) with different combinations of oxytocin, bolus and infusion, following caesarean section. The first trial9 found that significantly more women needed additional oxytocic interventions in the oxytocin group. The second trial10 found no significant differences in the intraoperative blood loss. However, neither study compared carbetocin directly with the currently recommended (and licensed) dose of oxytocin (5 IU) so we performed this comparison in a double-blind randomised trial.
Methods
Setting
The study took place in Southmead Hospital which is a teaching hospital in Bristol, UK. There are approximately 6000 deliveries per annum.
Study design
Double-blind randomised single centre study (1:1 ratio).
Primary outcome
The proportion of women in each arm of the trial that needed additional pharmacological oxytocic interventions.
Secondary outcomes
Estimated blood loss, difference in preoperative and post- operative haemoglobin, vital signs during and after the operation, uterine tone, incidence of blood transfusion and adverse effects.
Inclusion criteria
Women with a singleton pregnancy undergoing elective or emergency caesarean section after 37 weeks of gestation.
Exclusion criteria
Women with multiple gestation, placenta praevia and pla- cental abruption were excluded because there is a higher risk of haemorrhage with these conditions and it was therefore felt to be inappropriate to recruit these women. Women undergoing caesarean section with general anaes- thesia were also excluded, because carbetocin is licensed for use with regional anaesthesia only. Furthermore, we excluded women undergoing caesarean section at less than 37 weeks of gestation (likely to be emergency caesar- ean sections; a different smaller group from term preg- nancies) and women having emergency caesarean section for fetal or maternal distress where, due to time con- straints, it was not possible and/or appropriate to recruit or randomise.
Sample size estimation
A previous audit (2004) in our hospital (Southmead Hospi- tal, North Bristol NHS Trust) showed that additional oxytoc- ics were used after 21% of all caesarean sections. We hypothesised that carbetocin could halve this figure to 10%, which would be a clinically significant finding (in the Canadian multicentre study9 carbetocin reduced the use of addi- tional oxytocics from 10.1 to 4.7%). Power calculation was performed with SigmaStat version 2.03 (SPSS Inc, Chicago, IL, USA). We calculated that we needed a sample size of 338 women (169 in each arm) to detect this difference (21% versus 10%, power 80%, a = 0.05). We decided to recruit 190 women in each arm (total = 380) to allow for partici- pant withdrawal, mistakes in randomisation etc.
Recruitment and consent
Women undergoing elective caesarean sections were approached and recruited in the antenatal clinic. In addi- tion, we recruited women undergoing induction of labour and women in early labour with risk factors for caesarean section (e.g. previous caesarean section, macrosomia etc.). Women in early labour were invited to participate, only if there was adequate time to consider their participation (at least 4 hours) and they were not too distressed from labour (e.g. epidural analgesia). Signed, informed consent was obtained from all women at the point of recruitment to the study.
Randomisation
The randomisation sequence (1:1 ratio—blocks of ten, no stratification) was generated by computer. Women rando- mised in the study would receive either 5 IU oxytocin (SyntocinonÒ; Alliance, Chippenham, UK) or 100 lg car- betocin (PabalÒ; Ferring, Langley, UK). The preparation of the ‘blinded’ ampoules was undertaken by DHP Ltd. (Powys, UK) which provided 380 sequentially numbered and labelled boxes each containing a 1-ml ampoule of the study drug (either oxytocin or carbetocin according to the randomisation order) with an ampoule snapper already placed on top of the ampoule, to reduce the likelihood of an ampoule breaking. All boxes and ampoules were identi- cally labelled, with the study number (1–380) being the only differentiating feature between different drug packs. The random allocation sequence was not known to the investigators until the study had finished and the analysis was started. Recruited women were randomised into the study immediately before transfer to theatre, when the next consecutively numbered box was taken to theatre for administration of the study medication after the birth of the neonate.
Study drug administration
The study medication (carbetocin or oxytocin) was diluted in 10 ml normal saline and administered slowly (over 30– 60 seconds) intravenously by the anaesthetist after the birth of the baby. The slow administration has been shown to reduce the potentially harmful haemodynamic effects of oxytocin11,12 (and presumably carbetocin). Peri- operative and postoperative care was otherwise normal.
Administration of additional oxytocics was at the discretion of the operating surgeon. There was no departmental guide- line at the time of the study describing risk factors when additional prophylactic oxytocics should be given.
Data collection
Demographic, pregnancy and postnatal data were recorded by the researchers on the study proformas. Data relating to the operation [indication, estimated blood loss, additional oxytocic(s) used, uterine tone on a scale 1–10 and adverse effects] were recorded on proformas filled in by the operat- ing obstetrician. Blood loss was estimated by the surgeon in the usual way (visual estimation, number of used swabs and amount of aspirated blood). Blood pressure and pulse readings were recorded on the anaesthetic and recovery charts. Women were followed up to discharge from the hospital.
Statistical analysis
The data were analysed by intention to treat. Simple group comparisons were made using chi-square tests (with conti- nuity corrections in the case of 2 · 2 tables) and Student’s t tests for categorical and continuous variables respectively. Mann–Whitney U tests were used for variables that were not normally distributed. Mean profiles of blood pressure and pulse rates up to 40 minutes after the administration of the study drug were compared with repeated measures Analyses of Variance, using the ‘proc MIXED’ facility implemented in the sas software package (SAS version 9.1;SAS Inst. Inc., Cary, NC, USA, 2002/3). A 5% level of sig- nificance was used throughout.
Ethics
Ethical approval was granted by the Newcastle 1 Research Ethics Committee (reference: 05/Q0905/137). The study was registered with the European Clinical trials database (EudraCT number: 2005-002812-94). We followed the CONSORT statement for the reporting of the study.13
Results
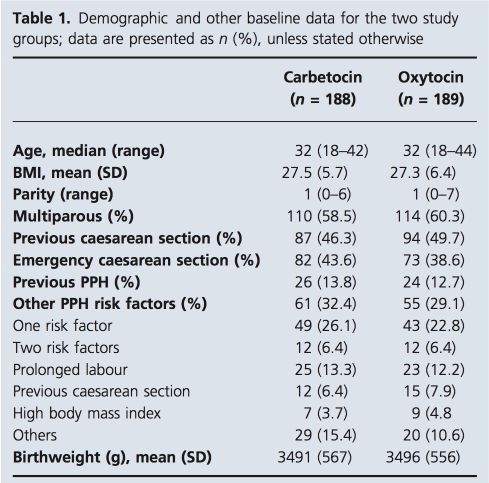
Recruitment and randomisation took place between November 2006 and July 2007. The randomisation flow- chart is demonstrated in Figure 1. Approximately 40% of invited women were eventually randomised in the study; 26% of the invited women declined participation and 34% did not have a caesarean section, although they had entered the study and would have been randomised, if they had needed abdominal delivery. There were three randomisa- tion errors in total and in all of these the next consecutive pack was opened. A total of 377 women were randomised in the study and analysed. Baseline demographic data for the two study groups were broadly similar and are pre- sented in Table 1. Approximately 40% of the participants had emergency caesarean sections and more than half of them were multiparous. Furthermore, almost one-third of women had previous PPH or other PPH risk factors and the proportions were similar between the two groups.
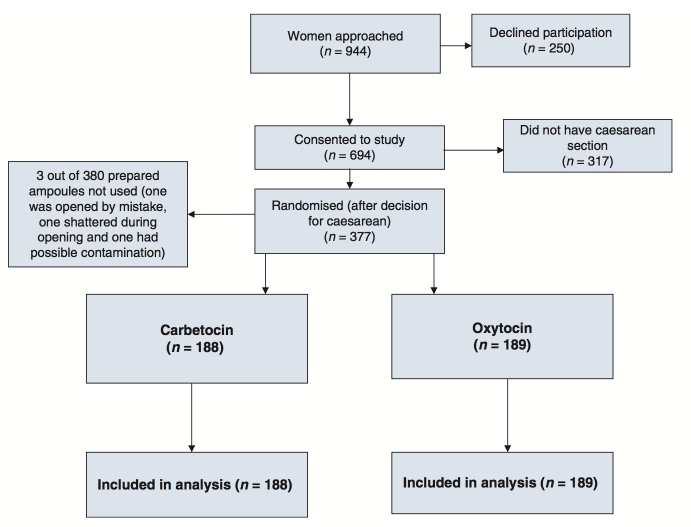
Figure 1. Randomisation flowchart.
The results for the main outcomes and secondary out- comes are presented in Table 2. More than one-third of women in the study required additional oxytocics. 33.5% of women in the carbetocin group needed additional oxy- tocics versus 45.5% of women in the oxytocin group [rela- tive risk (RR) 0.74, 95% confidence interval (95% CI) 0.57–0.95, P = 0.023). Therefore, significantly more women required additional oxytocics in the oxytocin group. The majority of these women had 40 IU oxytocin infusions, which were typically administered over 4 hours. The sur- geons documented in the study proforma whether the additional oxytocics were given for PPH treatment (as per- ceived by the surgeon), or prophylaxis. They quoted treat- ment of PPH as the indication for additional oxytocic administration more frequently in the oxytocin group and this difference was statistically significant. There were no significant differences in the estimated blood loss, uterine tone at the end of the operation, number of women with major PPH (blood loss >1000 ml) or number of women requiring blood transfusions. Similarly there were no signif- icant differences in the mean haemoglobin fall after the
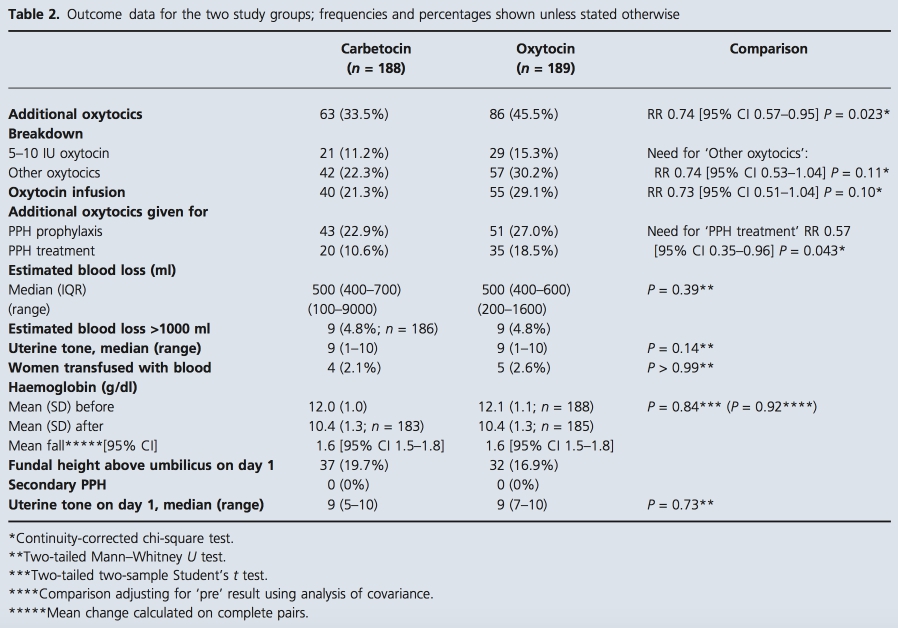
operation and in the fundal height or uterine tone postna- tally.
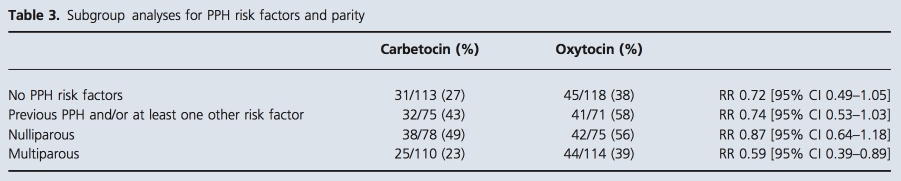
Subgroup analyses for the presence of risk factors, including previous PPH, as well as for parity are presented in Table 3. The use of additional oxytocics was reduced in the carbetocin arm, irrespective of the presence of risk fac- tors for PPH. Interestingly, there was no significant differ- ence in the use of additional oxytocics for nulliparous women (high usage of additional oxytocics for both study arms), with carbetocin mainly reducing the use of addi- tional oxytocics in multiparous women. However, in a logistic regression model (relating the log odds of addi- tional oxytocin to treatment group and parity), the interac- tion between group and parity was not statistically significant (P = 0.29).
There was one woman in the carbetocin arm who suf- fered a massive PPH following caesarean section for delay in the first stage of labour. Her total estimated blood loss was 9000 ml and she received a transfusion of 14 units of
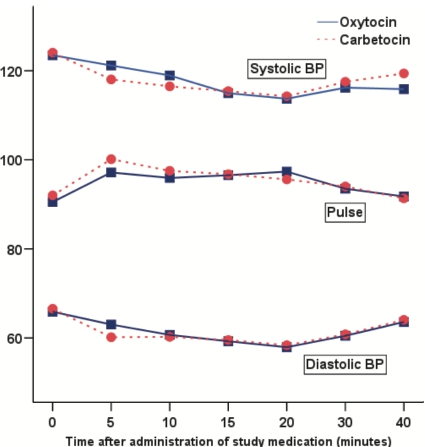
Figure 2. Mean systolic blood pressure (BP), diastolic BP and pulse for the first 40 minutes after administration of the study medication.
red blood cells. In addition to carbetocin, two doses of erg- ometrine, 800 lg misoprostol, 1250 lg carboprost and a 4-hour infusion of 40 IU oxytocin were administered. The uterine arteries were ligated and a caesarean hysterectomy was avoided.
Figure 2 demonstrates the mean systolic blood pressure, diastolic blood pressure and pulse up to 40 minutes after the drug administration. There are significant changes with time (P < 0.001 for each of systolic blood pressure, dia- stolic blood pressure and pulse, respectively) but no signifi- cant main effect of group (P = 0.77, P = 0.57 and P = 0.77, respectively).
Table S1 summarises the reported adverse effects of the two interventions. The adverse effect profile appears similar and there was no significant difference in the number of women affected by at least one adverse effect.
Discussion
This is the first study, to the best of our knowledge, to compare carbetocin with the licensed dose of oxytocin. The results demonstrate an increased use of additional oxytocics in the oxytocin arm. Most of the women who were given additional oxytocics received additional oxytocin bolus or infusion. Almost one in three women in the oxytocin arm received an additional oxytocin infusion, which was typi- cally given over 4 hours. The reason for administering additional oxytocics in the oxytocin group (according to the surgeon) was significantly more likely to be PPH treat- ment.
The results suggest that carbetocin may be a more potent oxytocic, but it is unclear whether this will reduce the rate of PPH and in particular major PPH. Certainly, there was no significant difference in the estimated blood loss, although this can be imprecise,14 especially for blood loss more than 600 ml.15 There was also no difference in the postoperative fall in haemoglobin, although this can be an imprecise blood loss measure. Reassuringly, more than 90% of women in both groups had their haemoglobin checked on the second postoperative day, as per depart- mental protocol. All the previous studies of carbetocin9,10 demonstrated a lower rate of additional oxytocic usage, but no study (including this one) has demonstrated a significant difference in the rate of PPH, which is arguably a more important outcome. The reason for this is that only a very large study with many thousands of women would have adequate power to demonstrate a significant difference in this relatively rare outcome. Perhaps, large retrospective studies from countries or institutions where carbetocin is used routinely may provide interesting data, although such studies would be prone to bias.
Nevertheless, the lower use of additional oxytocics is an important outcome with possible financial savings if the additional oxytocics require prolonged administration on the labour ward or in the recovery area. However, this may be offset by the higher cost of carbetocin in comparison to oxytocin. The UK cost of a carbetocin ampoule (one dose) is £17.64 (although a reduced price may be available), whereas the cost of a 10 IU ampoule of oxytocin is £0.86.
The haemodynamic data are reassuring with no clinically significant differences between the two interventions. How- ever, many of the haemodynamic changes resulting from oxytocin occur within the first 5 minutes12 and our study outcomes did not include continuous haemodynamic mon- itoring during this time period, as it was previously dem- onstrated9 that the two drugs had similar haemodynamic profiles. Although, the slow intravenous administration of oxytocics appears to reduce their haemodynamic effects,12 further studies comparing the haemodynamic profiles of carbetocin and oxytocin by invasive monitoring with arte- rial line will provide more robust data on this subject.
Overall, the adverse effect profiles appear reassuringly similar between the two medications. It could be argued that some of these are not ‘true’ adverse effects, but rather are the effect of hypotension or surgery.
We minimised bias by using a double-blind randomised design and widened the external validity of the results by including emergency caesarean sections in the study popu- lation. We achieved a low rate of randomisation errors with complete primary outcome data for all randomised women. However, we did not collect data for the 26.5% of invited women who declined participation, and therefore we could not compare their characteristics to the women who accepted. Overall, the women who were randomised included 37% of all caesarean sections that happened dur- ing the study period, suggesting that the study population should be reasonably representative of the caesarean section population.
However, a potential drawback of the study was that the use of additional oxytocics was unusually high in the study population. The overall use of additional oxytocics approached 40% in the study, compared with the 20% use in the previous audit, which was completed 12 months ear- lier. It is difficult to explain this difference in such a short time, but it may demonstrate a lower threshold by less senior obstetricians for giving additional oxytocics (usually oxytocin infusions) for PPH prophylaxis. Another possible explanation is that almost one-third of our study popula- tion had at least one risk factor for PPH. In a recent survey of practice in the UK,7 approximately 20% of clinicians reported routine use of oxytocin infusions whereas 80% reported selective use in the presence of risk factors. In our study, more than 50% of the additional oxytocic drugs were given for PPH prophylaxis according to the operating surgeon. Nevertheless, the subgroup analysis in Table 3 demonstrates that carbetocin reduces the use of additional oxytocics irrespective of the presence of risk factors for PPH.
The second subgroup analysis showed no significant dif- ference in the primary outcome for nulliparous women, probably because the use of oxytocics was high in both groups (almost 50%). This is likely to be a result of the higher proportion of emergency caesarean sections among nulliparous women. It is notable that the effect of carbeto- cin was more obvious in multiparous women (RR 0.59), but these subgroup analyses should be interpreted cau- tiously because the numbers involved are unavoidably small.
The high use of additional oxytocics (which was an infu- sion of oxytocin for the majority of women) raises the question whether an additional oxytocin infusion should be administered routinely after a caesarean section, given the short half-life of oxytocin. This is currently the subject of a randomised trial.16 If the oxytocin infusion proves superior to the oxytocin bolus, the next logical step would be to compare carbetocin with oxytocin bolus followed by oxyto- cin infusion. Such a study should include in the outcomes the duration of stay in the Delivery Suite/Recovery area, as this has an impact on the efficiency of busy maternity units.
It is not known whether the carbetocin data from caesar- ean sections can be extrapolated to vaginal births. There has been one randomised study17 of carbetocin versus oxy- tocin following vaginal birth which showed an increased rate of additional uterotonic interventions (mainly uterine massage) in the oxytocin group. There have also been two randomised studies of carbetocin versus Syntometrine (Alli- ance) following vaginal birth,18,19 which did not demon- strate significant differences between the two groups in the use of additional oxytocics, fall in haemoglobin,18 incidence of PPH or estimated blood loss. However, both studies showed that carbetocin was associated with a significantly lower incidence of nausea and vomiting. The earlier study18 showed that carbetocin was associated with a lower inci- dence of hypertension at 30 and 60 minutes but a higher incidence of maternal tachycardia. These outcomes were not reported in the other study.19 Therefore, as for caesar- ean section, it is not possible to determine whether car- betocin reduces the incidence of PPH following vaginal delivery, possibly because all the studies have been under- powered for this rare outcome.
As carbetocin appears to have a similar haemodynamic profile to oxytocin (and both appear to have a more ‘benign’ adverse effect and haemodynamic profile than syn- tometrine), then carbetocin may become the drug of choice for women who have contraindications to the administra- tion of Syntometrine, such as women with hypertensive disorders of pregnancy and women with cardiac problems. Currently, pre-eclampsia and eclampsia are listed in the British National Formulary as contraindications to the administration of carbetocin. Cardiovascular disease is listed in the cautions with advice to avoid the drug’s use in severe cardiovascular disease. However, if the similarity of the haemodynamic profiles of the two drugs is confirmed in a study similar to the study by Thomas et al.12 (where arterial lines were used for continuous blood pressure mea- surement), then carbetocin would be preferable, as it has already been shown to reduce the use of additional oxytoc- ics and may also reduce the incidence of PPH. Such a study should be a priority.
In conclusion, carbetocin reduces the use of additional oxytocics following caesarean section when compared with the licensed dose of oxytocin (5 IU). If carbetocin is shown to have a similar haemodynamic profile to oxytocin, it may become the medication of choice for women with hyper- tensive disorders or cardiac problems. The question of whether carbetocin reduces the incidence of PPH is still unanswered.
Disclosure of interests
George Attilakos has received travel expenses from Ferring to present the study data in an international meeting. Jona- than Ash has received travel expenses from Ferring to pres- ent study data in a national meeting.
Contribution to authorship
GA conceived idea, performed literature review, designed study, recruited women, collected data, assisted with statis- tical analysis and authored the manuscript. DP recruited women, collected data and co-authored the manuscript. JA recruited women, collected data and co-authored the man- uscript. RB recruited women, collected data and co- authored the manuscript. CW recruited women, collected data and co-authored manuscript. FD designed study and co-authored the manuscript. LH performed statistical anal- ysis and co-authored the manuscript. TD conceived the idea, designed the study and co-authored the manuscript.
Details of ethics approval
Ethical approval was granted by Newcastle 1 Research Eth- ics Committee (reference: 05/Q0905/137) on 9 September 2005. Clinical trial authorisation was granted by the Medicines and Healthcare products Regulatory Agency (EudraCT number: 2005-002812-94).
Funding
Ferring UK funded the cost of preparation of the ‘blinded’ drug ampoules. No other external funding was required for the study.
Acknowledgements
We would like to thank all the midwives and doctors who helped with recruitment in the study. We would also like to thank all the women who took part in the study.
Supporting information
The following supplementary materials are available for this article:
Table S1. Reported side-effects in each group.
Additional Supporting Information may be found in the online version of this article.
Please note: Wiley-Blackwell are not responsible for the content or functionality of any supporting information supplied by the authors. Any queries (other than missing material) should be directed to the corresponding author.
References
1 Carroli G, Cuesta C, Abalos E, Gulmezoglu AM. Epidemiology of postpartum haemorrhage: a systematic review. Best Pract Res Clin Obstet Gynaecol 2008;22:999–1012.
2 Confidential Enquiry into Maternal and Child Health. Why Mothers Die 2000–2002. London, UK: Royal College of Obstetricians and Gynaecologists, 2004.
3 Stones RW, Paterson CM, Saunders NJ. Risk factors for major obstet- ric haemorrhage. Eur J Obstet Gynecol Reprod Biol 1993;48:15–8.
4 Villar J, Valladares E, Wojdyla D, Zavaleta N, Carroli G, Velazco A, et al. Caesarean delivery rates and pregnancy outcomes: the 2005 WHO global survey on maternal and perinatal health in Latin Amer- ica. Lancet 2006;367:1819–29.
5 National Collaborating Centre for Women’s and Children’s Health. Intrapartum Care: Care of Healthy Women and their Babies During Childbirth. London: NICE, 2007.
6 National Collaborating Centre for Women’s and Children’s Health. Caesarean Section. London: NICE, 2004.
7 Wedisinghe L, Macleod M, Murphy DJ. Use of oxytocin to prevent haemorrhage at caesarean section – a survey of practice in the Uni- ted Kingdom. Eur J Obstet Gynecol Reprod Biol 2008;137:27–30.
8 Sweeney G, Holbrook AM, Levine M, Yip M, Alfreddson K, Cappi S, et al. Pharmacokinetics of carbetocin, a long-acting oxytocin ana- logue, in nonpregnant women. Curr Ther Res 1990;47:528–40.
9 Dansereau J, Joshi AK, Helewa ME, Doran TA, Lange IR, Luther ER, et al. Double-blind comparison of carbetocin versus oxytocin in pre- vention of uterine atony after cesarean section. Am J Obstet Gyne- col 1999;180:670–6.
10 Boucher M, Horbay GL, Griffin P, Deschamps Y, Desjardins C, Schulz M, et al. Double-blind, randomized comparison of the effect of carbetocin and oxytocin on intraoperative blood loss and uterine tone of patients undergoing cesarean section. J Perinatol 1998;18:202–7.
11 Weis FR Jr, Markello R, Mo B, Bochiechio P. Cardiovascular effects of oxytocin. Obstet Gynecol 1975;46:211–4.
12 Thomas JS, Koh SH, Cooper GM. Haemodynamic effects of oxytocin given as i.v. bolus or infusion on women undergoing Caesarean sec- tion. Br J Anaesth 2007;98:116–9.
13 Moher D, Schulz KF, Altman DG. The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomised trials. Lancet 2001;357:1191–4.
14 Khan FA, Khan M, Ali A, Chohan U. Estimation of blood loss during Caesarean section: an audit. J Pak Med Assoc 2006;56:572–5.
15 Duthie SJ, Ghosh A, Ng A, Ho PC. Intra-operative blood loss during elective lower segment caesarean section. Br J Obstet Gynaecol 1992;99:364–7.
16 Murphy DJ, Carey M, Montgomery AA, Sheehan SR. Study protocol. ECSSIT – Elective Caesarean Section Syntocinon Infusion Trial. A multi-centre randomised controlled trial of oxytocin (Syntocinon) 5 IU bolus and placebo infusion versus oxytocin 5 IU bolus and 40 IU infusion for the control of blood loss at elective caesarean sec- tion. BMC Pregnancy Childbirth 2009;9:36.
17 Boucher M, Nimrod CA, Tawagi GF, Meeker TA, Rennicks White RE, Varin J. Comparison of carbetocin and oxytocin for the pre- vention of postpartum hemorrhage following vaginal delivery: a double-blind randomized trial. J Obstet Gynaecol Can 2004;26: 481–8.
18 Leung SW, Ng PS, Wong WY, Cheung TH. A randomised trial of carbetocin versus syntometrine in the management of the third stage of labour. BJOG 2006;113:1459–64.
19 Su LL, Rauff M, Chan YH, Suphan NM, Lau TP, Biswas A, et al. Car- betocin versus syntometrine for the third stage of labour following vaginal delivery; a double-blind randomised controlled trial. BJOG 2009;116:1461–6.
