Carbetocin versus oxytocin for prevention of post-partum haemorrhage at caesarean section in the United Kingdom: an economic impact analysis

Author: Helen Avan der Nelson, Tim Draycott, Dimitrios Siassakos, Christopher W.H Yau, Anthony J. Hatswell
PII: S0301-2115(17)30002-7
DOI: http://dx.doi.org/doi:10.1016/j.ejogrb.2017.01.004
Reference: EURO 9738
To appear in: EURO
Received date: 4-8-2016
Revised date: 16-12-2016Accepted date: 2-1-2017
Please cite this article as: der Nelson Helen Avan, Draycott Tim, Siassakos Dimitrios, Yau Christopher WH, Hatswell Anthony J.Carbetocin versus oxytocin for prevention of post-partum haemorrhage at caesarean section in the United Kingdom: an economicimpact analysis.European Journal of Obstetrics and Gynecology and Reproductive Biology http://dx.doi.org/10.1016/j.ejogrb.2017.01.004
This is a PDF file of an unedited manuscript that has been accepted for publication.As a service to our customers we are providing this early version of the manuscript.The manuscript will undergo copyediting, typesetting, and review of the resulting proofbefore it is published in its final form. Please note that during the production processerrors may be discovered which could affect the content, and all legal disclaimers thatapply to the journal pertain.
Carbetocin versus oxytocin for prevention of post-partum haemorrhage at caesarean section inthe United Kingdom: an economic impact analysis
Dr Helen A van der Nelson1,2, Prof Tim Draycott1, Dr Dimitrios Siassakos1,2, Dr Christopher W H Yau1,Mr Anthony J Hatswell3,4
(1) Academic Centre for Women’s Health, North Bristol NHS Trust, Bristol, United Kingdom
(2) School of Clinical Sciences, University of Bristol, 69 St Michael’s Hill, Bristol, BS2 8DZ
(3) BresMed, 84 Queen Street, Sheffield, S1 2DW
(4) Department of Statistical Science, University College London, Gower Street, London,WC1E 6BT
Corresponding author: Dr Helen van der Nelson, Maternity Research Office, The Chilterns,Southmead Hospital, Southmead Road, Westbury-on-Trym, Bristol, BS10 5NB, UK. Tel: (+44) 0117414 6764. Email: helenvandernelson@gmail.com
Abstract
Objective
To determine the economic impact of the introduction of carbetocin for the prevention ofpostpartum haemorrhage (PPH) at caesarean section, compared to oxytocin.
Study design
The model is a decision tree conducted from a UK National Health Service perspective. 1500caesarean sections (both elective and emergency) were modelled over a 12 month period. Efficacydata was taken from a published Cochrane meta-analysis, and costs from NHS Reference costs, theBritish National Formulary and the NHS electronic Medicines Information Tool. A combination ofhospital audit data and expert input from an advisory board of clinicians was used to informresource use estimates. The main outcome measures were the incidence of PPH and total cost overa one year time horizon, as a result of using carbetocin compared to oxytocin for prevention of PPHat caesarean section.
Results
The use of carbetocin compared to oxytocin for prevention of PPH at caesarean section wasassociated with a reduction of 30 (88 vs 58) PPH events (>500ml blood loss), and a cost saving of£27,518. In probabilistic sensitivity analysis, carbetocin had a 91.5% probability of producing betteroutcomes, and a 69.4% chance of being dominant (both cheaper and more effective) compared tooxytocin.
Conclusion
At list price, the introduction of carbetocin appears to provide improved clinical outcomes alongwith cost savings, though this is subject to uncertainty regarding the underlying data in efficacy,resource use, and cost.
Keywords: Cost-effectiveness, Economic model, carbetocin, postpartum haemorrhage.
Introduction
Primary Post Partum Haemorrhage (PPH) is most commonly defined as blood loss of 500ml or morefrom the genital tract within 24 hours of childbirth(1). Uterine atony is the cause of up to 90% of PPHand is increasing(2). Prophylactic uterotonic drugs are part of the active management of the thirdstage of labour that reduces risk of PPH by 66% when compared with physiological management(3),and a World Health Organisation (WHO) study concluded that haemorrhage prevention programmesshould focus on the use of uterotonic drugs(4).
The National Institute for Health and Clinical Excellence (NICE) in the United Kingdom currentlyrecommend oxytocin (Syntocinon®, Alliance) as the uterotonic drug of choice for PPH prophylaxis: a10IU intramuscular dose for vaginal births(5) and a 5IU slow intravenous dose for caesareanbirths(6). Carbetocin (Pabal®, Ferring) is a synthetic analogue of oxytocin, with structuralmodifications that increase its half-life and duration of action(7). A Cochrane review(8) concludedthat use of carbetocin resulted in a statistically significant reduction in the use of additionaluterotonic drugs at caesarean section when compared with oxytocin and a numerical reduction inthe incidence of PPH. Although carbetocin is likely to be at least as clinically effective as oxytocin, itis more expensive, with little published evidence on the cost-effectiveness of its use – as highlightedby the Cochrane review(8). The data that does exist is conflicting and of variable quality (9-11).
In this paper we describe the use of a health economic model constructed to assess the costeffectiveness of carbetocin for PPH prophylaxis at caesarean section from the perspective of the UKNational Health Service.
Methods
A decision tree was constructed in Microsoft Excel 2010® to model prophylactic doses of 5 IUintravenous oxytocin, or a single prophylactic (100µg) dose of intravenous carbetocin at caesareansection for PPH prevention. Oxytocin and Carbetocin were selected for comparison as they representcurrent UK prophylactic uterotonic practice and a longer lasting and potentially more effective (butmore expensive) alternative, respectively. The evaluation was undertaken from a National HealthService perspective, in keeping with UK National Institute for Health and Care Excellencerecommendations(12).
The primary outcome measures were the number of PPH events prevented and the impact on totalcost incurred by a large maternity unit over a one year time horizon, as a result of using carbetocininstead of oxytocin for PPH prevention at caesarean section. The study population comprised allwomen undergoing elective and emergency caesarean section. The number of caesarean sectionsperformed in the model was set to 1500, based on a unit with approximately 6500 deliveries perannum (a caesarean section rate of about 24%). Hospital-level audit data was used to informestimates of resource use.
Treatment pathway
The modelled treatment pathway is shown in Figure 1. Patients undergoing caesarean sectionreceive a prophylactic uterotonic drug after their delivery. Despite this prophylaxis, some womenexperience uterine atony requiring additional uterotonic drugs that will prevent PPH in some, butnot all cases. Patients experience varying volumes of blood loss at caesarean section – in the modelthis is captured in 4 health states – ‘No PPH event’, ‘PPH 500-999ml’, ‘PPH 1000-1499ml’ and “PPH>1500ml”. Larger volumes of blood loss are associated with more treatment and resource use, andas a result are more expensive. Table 1 shows the inputs to the model by different levels of bloodloss.
Patients are monitored in recovery for 2 hours after their caesarean section, as recommended bynational guidelines(6). Patients requiring additional uterotonic drugs (e.g. 4 hour oxytocin infusion)stay in recovery, or on labour ward, for longer. In these areas staff to patient ratios are greater, andmore medical time is utilised. Patients who experience a large PPH are more likely to requirepostnatal follow up, and for their care to be discussed at a risk management forum. A combinationof published data and hospital-level data was used in the economic model (Table 2).
Clinical effectiveness
Relative clinical efficacy was obtained from a published Cochrane Collaboration meta-analysis of fourrandomised control trials of carbetocin and oxytocin for PPH prophylaxis at caesarean section(8).Data used included the rate of PPH and the proportion of patients requiring additional uterotonicdrugs (Table 3). Point estimates (published means) have been used in the base case, withprobabilistic estimates also presented, as the Cochrane review concluded that although thereduction in the use of additional uterotonic drugs was statistically significant, the estimate fordifferences in PPH rates was not (p=0.086). The use of values (even if statistically insignificant), witha confidence interval around their estimates is well established in health economics(13), as suchestimates provide a the best estimate to the real world, where evidence is not always clear and isassociated with uncertainty regardless of the significance of the finding.
During the review of existing literature, we noted that the reporting of PPH categories isinconsistent: the meta-analysis data(8) includes analyses of both “PPH >1000ml” (two studies), and“PPH >500ml or as defined by trialist” (all four studies). To account for incomplete reporting, theproportion of total PPH events in each blood loss category was interpolated from a cohort study of1584 women in the Netherlands, which compared carbetocin and several different dosing regimensof oxytocin for PPH prophylaxis during caesarean section(14). Data regarding the distribution of PPHevents across these categories was provided by the publishing authors. The resulting data for PPH ineach blood loss category for all efficacy sources is shown in Table 1, with the distribution ofoutcomes assumed to be the same in both arms.
Resource use
Clinical management and resource use escalates with increasing blood loss. The proportion of casesrequiring additional uterotonic drugs was derived from clinical effectiveness data shown in Table 3.The additional uterotonic drug assumed was 5 IU oxytocin given by slow IV bolus, representing UKrecommended clinical practice(15). This single dose was assigned to patients needing “additionaluterotonics”, as not all patients who require additional uterotonic drugs go on to experience a PPH.The resources required to manage a PPH in each of the categories used in the economic model (500-999ml, 1000-1499ml and ≥1500ml), was estimated by a multi-professional panel of clinical experts.Resource use included the type (and number of doses) of additional drugs needed, as well as stafftime associated with treatment of the PPH. The resulting assumptions are shown in Table 2. Theseare in line with national guidance on the management of PPH(15).
Where published estimates were not available, hospital level data (from Southmead Hospital,Bristol, UK) was used to improve accuracy of resource use estimates. Hospital level data includedaverage length of maternal inpatient stay post caesarean, proportion of patients needing a bloodtransfusion, units of red blood cells transfused, and provision of consultant follow-up in the first tenpostnatal weeks. Each item was also calculated for blood loss in the ranges 0-499ml (no PPH), 500-999ml, 1000-1499ml and ≥1500ml (see Table 3).
Unit cost estimates
Costs were calculated in Pounds Sterling, and were taken from NHS Reference costs, the BritishNational Formulary and the NHS electronic Medicines Information Tool (which contains the meanprice paid for generic pharmaceuticals in the UK).
Utilities
To provide a common unit of comparison, utilities decrements were used for the differing levels ofPPH. As no direct utility values were available, estimates for the disutility of gastrointestinal bleeds(16) were used, such as a disutility of 0.06 for 7 days for PPH 500-1000ml, a disutility of 0.25 for 10days for PPH 1000-1500ml, and a disutility of 0.25 for 14 days for PPH>1500ml.
Results
Table 4 contains a breakdown of costs associated with the use of oxytocin or carbetocin forprevention of PPH at caesarean section in this model. In the base case, the use of carbetocin shows areduction of 30 PPH events (58 vs 88) and an estimated cost saving of £27,518.41 (£2,085,989 vs£2,113,508). This difference is mainly driven by a reduction in the number of PPH events(incremental cost saving £35,985) and the resultant reduction in time spent in recovery aftertreatment of PPH (incremental cost saving of £12,783). These savings offset the increased drug costof carbetocin compared to oxytocin (unit price £17.64 v £0.80, which gives an increase of £22,860per year).
Probabilistic sensitivity analysis, which takes into account the uncertainty in input values (bothclinical and cost) shows carbetocin to be more effective than oxytocin in 91.5% of scenarios, anddominant (both cheaper and more effective) in 69.4% of scenarios. When attaching utility values toPPH events based on assumed disutilities, carbetocin is cost-effective at a threshold of £20,000 perQALY in 70.5% of scenarios (Figure 2).
Discussion
Main findings
The model demonstrates that carbetocin is likely to provide superior clinical outcomes (by reducingthe rate of PPH events), along with a cost saving. However probabilistic analysis illustratesuncertainty due to the underlying data, where carbetocin does not provide cost savings (30.4%), anddoes not show cost-effectiveness using the NICE threshold for recommendation (over £20,000 perQALY gained, 29.5% of scenarios).
Strengths and limitations of the study
The treatment pathway used in the model is in line with national guidelines(5, 6, 15). The resultsshould be applicable to most maternity units in the United Kingdom, regardless of size, as well asthose internationally with similar care pathways. Whilst UK guidelines recommend a single 5IUoxytocin dose for PPH prophylaxis, we are aware that practice varies greatly(17, 18), and that thereis some evidence suggesting that the addition of a postoperative oxytocin infusion may furtherreduce risk of PPH(19) ,as is common in other countries(20). This does not affect our conclusionshowever; using a 40IU infusion over 4 hours for all oxytocin patients increased the cost saving ofcarbetocin to £31,118 (an increase of approximately £3,600).
Our analysis is primarily influenced by the clinical effectiveness data chosen for each scenario.Cochrane meta-analysis data was used to inform the base case, which raises the question of themethodological differences and clinical heterogeneity between studies. An important difference wasthe variability in the method of IV oxytocin administration in each individual study (21-24). Eachstudy referred to their method of oxytocin infusion as “standard”, suggesting that the routine doseand administration method for prophylactic oxytocin differs between settings, and over time.Ultimately, a meta-analysis provides the best available clinical evidence whilst also reflecting someof the variability in clinical practice. A similar limitation is that adverse events were not included inthe model; in the absence of a definitive head to head trial, the variation in reporting and treatmentof adverse events in heterogeneous trials would introduce a bias of unknown magnitude anddirection. This is particularly the case as the treatments are given alongside a complex pathway ofinterventions; isolating the adverse events of PPH prophylaxis would be extremely difficult.
Although necessary for resource use analysis, the sub-categorisation of PPH by blood volume doesplace arbitrary limits on a continuous outcome. As such, the categories have been created based onthe literature available, and resources assigned to reflect the mean for patients falling within eachcategory. We are mindful that exceedingly large PPHs may incur additional costs such as use ofFactor VII, admission to Intensive Care, and potentially medico-legal expenses associated withlitigation, however no data exist on these rare events and as such they have been omitted from ourestimates so as not to bias the analysis. A further assumption in the model is that the breakdown ofPPH events into the different categories is assumed to be identical between the two treatments (asis the proportion of patients with additional PPH risk factors such as obesity, prolonged labour andplacenta praevia). This is representative of clinical practice – PPH is not treated differently based onthe prophylactic treatment received.
The lack of universally adopted categories of PPH that clinical trials use to classify obstetric bloodloss also causes uncertainty in the clinical data, which is carried through to modelling. Although themost commonly used definition of PPH is that quoted by the WHO (loss of >500ml blood from thegenital tract within 24 hours of birth), the outcomes reported in clinical trials vary considerably,particularly for trials involving caesarean section. As trials commonly only report blood loss in one ofthese categories (e.g. “PPH >1000ml”, or PPH “>500ml”), missing category data was accounted forby the interpolation of categorical PPH proportions from a large published dataset (14). These dataare from a large study population in a healthcare system comparable with that of the UK. However,this dataset only included elective caesarean sections, whereas PPH, particularly severe PPH, is morecommon following emergency caesarean section(25-27).
Interpretation in light of existing literature
A small study (11) performed a financial evaluation alongside a departmental audit in a UK hospital,after changing from routine use of oxytocin to carbetocin at elective caesarean section. This was anobservational study which only contained 24 patients in the oxytocin arm, and 37 patients in thecarbetocin arm. It concluded that carbetocin was associated with a £18.52 increased cost perpatient, and no significant clinical benefit. In addition to the small sample size and lack of formalmethods for adjusting for difference in baseline patient characteristics, it is not clear how these costswere estimated, and no formal economic modelling was performed. Similarly a Mexican abstract(9)reported an economic evaluation of carbetocin for the prevention of uterine atony in patients withrisk factors for PPH. This compared carbetocin with oxytocin, and included a total of 152 patients.Mode of delivery was not stated, nor were details of any blinding, costs included, or treatmentpathways used. It concluded that the overall cost per patient treated with carbetocin was 529 USDless than those treated with oxytocin (approximately £339 per patient). However without further information about the study, resource use, or the costs included, comparison with our results is notmeaningful.
A more useful comparison is with a cost-minimization analysis performed from a Canadianhealthcare system perspective(10) which investigated the use of carbetocin for prevention of PPHduring elective caesarean section, with treatment pathways modelled on guidelines from the Societyof Obstetricians and Gynecologists of Canada. This compared carbetocin with unnamed“comparators most commonly encountered in clinical practice”. Rather than using clinical trial datathe study “assumed that the incidence of PPH was equal between treatment strategies”, which willhave heavily influenced results and is inconsistent with economic evaluation guidelines(13). Thisanalysis reported a per patient cost of $31.95 for carbetocin vs $32.31 for oxytocin. While theseresults are more consistent our results, it is again difficult to draw comparisons due to the limitedinformation presented, and assumption of equal efficacy in prevention of PPH (contrary to publishedmeta-analytic data).
Conclusions
This economic evaluation combines the best available clinical effectiveness data for the use ofoxytocin versus carbetocin during caesarean section for PPH prophylaxis, with UK hospital-levelresource use data. The model estimates that carbetocin is likely to result in better clinical outcomesand a modest cost-saving when compared to oxytocin, albeit with substantial uncertainty.
Whilst the results of this model will help to inform policy makers, further work is needed. Thecurrent data indicate carbetocin is more effective than oxytocin in reducing the use of additionaluterotonic drugs, and although existing data indicates a numerical advantage for carbetocin inreducing the rate of PPH, this does not reach statistical significance. There exists thereforeuncertainty in the relative clinical effectiveness of carbetocin, which we hope will be provided by anongoing randomised control trial (“The IMox Study”, Clinicaltrials.gov NCT02216383).
Although carbetocin appears to have a number of advantages in our study (potentially includingcost), a large randomised trial of the use of these drugs at caesarean section with parallel healtheconomic evaluation, is required to conclusively inform practice. Until this has been conducted, theevaluation presented here uses the most robust information available and demonstrates that theintroduction of carbetocin is likely to result in better clinical outcomes and potentially a modest perpatient cost-saving, albeit with uncertainty. At the very least, it appears that carbetocin use wouldbe cost-neutral, meaning decisions regarding its introduction should be based on clinicaleffectiveness.
Disclosure and contribution of authorship.
The project was led by HvdN, the model was developed by AJH, clinical input was provided by HvdN,TD and DS. HvdN is the lead investigator on a clinical study that has received start up funding fromFerring (the manufacturers of carbetocin), TD has attended advisory board meetings sponsored byFerring. DS has attended expert meetings organised by Ferring and has organised events sponsoredby them. AJH is an employee of BresMed, who have received funding from both Ferring and Pfizer(the manufacturers of carboprost). All authors read and approved the final manuscript.
Funding:
No funding was received for this project.
Detail of ethical approval:
Ethical approval was not required for this project.
Acknowledgements:
The authors would like to acknowledge the help of Ash Bullement, Dawn Lee and Thea Henry inconstructing the economic model, Eva Creutzberg for provision of PPH data, advisory boardparticipants for clinical input on the management of PPH, and Helen Fenn for the collection ofresource use data from Southmead hospital, Bristol.
References
- WHO recommendations for the prevention and treatment of post partum haemorrhage.World Health Organisation. 2012. [Internet - last accessed 16/12/2016] Available from http://www.who.int/reproductivehealth/publications/maternal_perinatal_health/9789241548502/en/
- Knight M CW, Berg C, Alexander S, Bouvier-Colle M-H, Ford JB, et al. Trends in postpartumhemorrhage in high resource countries: a review and recommendations from the International Postpartum Hemorrhage Collaborative Group. BMC Pregnancy Childbirth. 2009;9(1):55.
- Begley CM, Gyte GM, Devane D, McGuire W, Weeks A. Active versus expectant managementfor women in the third stage of labour. Cochrane Database Syst Rev. 2015(3) Art. No.: 007412.
- Gulmezoglu AM, Lumbiganon P, Landoulsi S, Widmer M, Abdel-Aleem H, Festin M, et al.Active management of the third stage of labour with and without controlled cord traction: arandomised, controlled, non-inferiority trial. Lancet. 2012 May 5;379(9827):1721-7.
- Intrapartum Care for Healthy Women and Babies National Institute for Health and CareExcellence. Clinical Guideline CG 190. 2014. [Internet - last accessed 16/12/2016] Available from https://www.nice.org.uk/guidance/cg190
- Caesarean Section. National Institute for Health and Care Excellence. Clinical Guideline CG132. 2011. [Internet - last accessed 16/12/2016] Available from https://www.nice.org.uk/guidance/cg132
- Rath W. Prevention of postpartum haemorrhage with the oxytocin analogue carbetocin.European journal of obstetrics, gynecology, and reproductive biology. 2009 Nov;147(1):15-20.
- Su LL, Chong YS, Samuel M. Carbetocin for preventing postpartum haemorrhage. CochraneDatabase Syst Rev. 2012(4) Art. No.: 005457.
- Del-Angel-Garcia G, Garcia-Contratres F, Nevarez-Sida A, Constantino-Casas P. A costeffectiveness study of Carbetocine compared to oxytocin for the prevention of uterine atony inpatients with risk factors. Value in Health. 2006;9(3):A50.
- Millsa F CC. A cost-minimization analysis of Carbetocin for the prevention of post partumhemorrhage in Canada. Value in Health. 2014; 17(3):A161
- Higgins L, Mechery J, Tomlinson AJ. Does carbetocin for prevention of postpartumhaemorrhage at caesarean section provide clinical or financial benefit compared with oxytocin?Journal of Obstetrics and Gynaecology. 2011;31(8):732-9.
- Guide to the methods of technology appraisal. National Institute for Health and CareExcellence. Process and Methods PMG9. 2013. [Internet - last accessed 16/12/2016]. Available from https://www.nice.org.uk/process/pmg9
- Claxton K. The irrelevance of inference: a decision-making approach to the stochasticevaluation of health care technologies. Journal of Health Economics. 1999;18(3):341-64.
- Holleboom CA, van Eyck J, Koenen SV, Kreuwel IA, Bergwerff F, Creutzberg EC, Bruinse H.Carbetocin in comparison with oxytocin in several dosing regimens for the prevention of uterineatony after elective caesarean section in the Netherlands. Archives of Gynecology and Obstetrics.2013;287(6):1111-7.
- Prevention and management of post partum haemorrhage. Royal College of Obstetriciansand Gynaecologists. Greentop Guideline 52. 2016. [Internet - last accessed 16/12/2016]. Availablefrom https://www.rcog.org.uk/en/guidelines-research-services/guidelines/gtg52/
- Lee D, Thornton P, Hirst A, Kutikova L, Deuson R, Brereton N. Cost effectiveness ofromiplostim for the treatment of chronic immune thrombocytopenia in Ireland. Applied Health Economics and Health Policy. 2013;11(5):457-69.
- Wedisinghe L, Macleod M, Murphy DJ. Use of oxytocin to prevent haemorrhage at caesareansection--a survey of practice in the United Kingdom. European Journal of Obstetrics, Gynecology, and Reproductive Biology. 2008;137(1):27-30.
- Stephens LC, Bruessel T. Systematic review of oxytocin dosing at caesarean section.Anaesthesia and Intensive Care. 2012;40(2):247-52.
- Murphy DJ, MacGregor H, Munishankar B, McLeod G. A randomised controlled trial ofoxytocin 5IU and placebo infusion versus oxytocin 5IU and 30IU infusion for the control of blood lossat elective caesarean section--pilot study. ISRCTN 40302163. European Journal of Obstetrics,Gynecology, and Reproductive Biology. 2009;142(1):30-3.
- Mockler JC, Murphy DJ, Wallace EM. An Australian and New Zealand survey of practice ofthe use of oxytocin at elective caesarean section. The Australian & New Zealand Journal of Obstetrics& Gynaecology. 2010;50(1):30-5.
- Dansereau J, Joshi AK, Helewa ME, Doran TA, Lange IR, Luther ER, et al. Double-blindcomparison of carbetocin versus oxytocin in prevention of uterine atony after cesarean section. American Journal of Obstetrics and Gynecology. 1999;180(3 Pt 1):670-6.
- Boucher M, Horbay GL, Griffin P, Deschamps Y, Desjardins C, Schulz M, et al. Double-blind, randomized comparison of the effect of carbetocin and oxytocin on intraoperative blood loss anduterine tone of patients undergoing cesarean section. Journal of Perinatology. 1998; 18(3):202-7.
- Attilakos G, Psaroudakis D, Ash J, Buchanan R, Winter C, Donald F, et al. Carbetocin versusoxytocin for the prevention of postpartum haemorrhage following caesarean section: the results of adouble-blind randomised trial. BJOG. 2010;117(8):929-36.
- Borruto F, Treisser A, Comparetto C. Utilization of carbetocin for prevention of postpartumhemorrhage after cesarean section: a randomized clinical trial. Archives of Gynecology andObstetrics. 2009;280(5):707-12.
- S Daniel MV, B Simi, A Nazeema. Study of maternal outcome of emergency and electivecaesarean section in a semi-rural tertiary hospital. National Journal of Medical Research.2014;4(1):14-8.
- 26. Ghazi A, Karim F, Hussain AM, Ali T, Jabbar S. Maternal morbidity in emergency versuselective caesarean section at a tertiary care hospital. Journal of Ayub Medical College.2012;24(1):10-3.
- Holm C, Langhoff-Roos J, Petersen KB, Norgaard A, Diness BR. Severe postpartumhaemorrhage and mode of delivery: a retrospective cohort study. BJOG. 2012;119(5):596-604.
Figure 1: Treatment pathway
Figure 2: Scatterplot of Monte-Carlo estimates (1000 simulations)
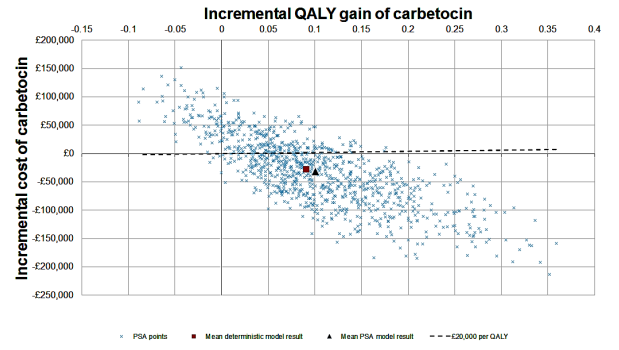
Table 1: Proportionate spread of PPH events across categories of blood loss
|
|
Proportion of all PPHs falling in each category |
Total |
||||
|
No PPH (blood loss <500ml) |
500- 999ml |
1000 – 1499ml |
≥1500ml |
|||
|
|
||||||
|
Holleboom 2013 |
Oxytocin |
680 |
319 |
66 |
57 |
1122 |
|
Carbetocin |
248 |
165 |
33 |
16 |
462 |
|
|
Raw data from published literature |
|
|||||
|
Su 2012 |
Oxytocin |
563 |
|
35 |
|
598 |
|
|
Carbetocin |
574 |
|
23 |
|
597 |
|
Model inputs, including interpolation of missing data using data from the Holleboom et al., dataset |
|
|||||
|
Su 2012 |
Oxytocin |
94.2% |
4.3% |
0.9% |
0.7% |
100% |
|
Carbetocin |
96.2% |
2.8% |
0.6% |
0.4% |
100% |
|
Table 2: Cost and resource use inputs for PPH events
|
|
Resources needed for management of PPH in each category |
Source of resource use estimate |
|||
|
PPH 500- 999ml |
PPH 1000- 1499ml |
PPH ≥1500ml |
|||
|
Additional uterotonic drugs given to treat PPH (cost taken from BNF volume 67) |
|||||
|
Oxytocin 5iU (Syntocinon®, £0.80) |
1 |
1 |
1 |
Expert opinion (see footnote) |
|
|
Ergometrine 0.5IU and Oxytocin 1mL (Syntometrine®, £1.35) |
0 |
1 |
1 |
Expert opinion |
|
|
Carboprost 250µg (Haemobate® £18.20) |
0 |
1 |
3 |
Expert opinion |
|
|
Intravenous replacement (cost take from sources as stated) |
|||||
|
Hartmann’s solution 500ml IV (£2.75 – Just Care Medical) |
1 |
3 |
5 |
Expert opinion |
|
|
Red blood cells, 1 unit (£122.09 – NHS Blood & Transfusion Services) |
1.5%, mean 2.3 units |
7.5%, mean 2.1 units |
23%, mean 2.6 units |
NHS Trust data |
|
|
Fresh Frozen Plasma, 1 unit (£33.81 – NHS Blood & Transfusion Services) |
0 |
0 |
2 |
Expert opinion |
|
|
Hours of staff time needed, in addition to routine uncomplicated caesarean section (costs taken from PSSRU 2013) |
|||||
|
Anaesthetist (£94/hour) |
0 |
0 |
4 |
Expert opinion |
|
|
Obstetrician (£100/hour) |
0 |
0 |
4 |
Expert opinion |
|
|
Midwife (£65.44/hour) |
0 |
0 |
4 |
Expert opinion |
|
|
Junior doctor (£29/hour) |
0 |
0 |
4 |
Expert opinion |
|
|
Haematologist (£99/hour) |
0 |
0 |
1 |
Expert opinion |
|
|
Other costs (costs taken from NHS Reference Costs 2012/2013) |
|||||
|
Additional length of post-natal inpatient stay (days), £439.35/day |
0.03 |
0.28 |
1.19 |
NHS Trust data |
|
|
Days required in high dependency unit such as 1:1 care on Labour Ward (£630/day) |
0 |
1 |
1 |
Expert opinion |
|
|
Additional cost for operating theatre per hour (£1139.60 – ISD Scotland) |
0 |
1 |
1 |
Expert opinion |
|
|
Case discussion at PPH meeting(£67.14) |
0 |
0 |
1 |
Expert opinion |
|
|
Post natal consultant follow up in first 10 post natal weeks (£129.92) |
2.9% |
2.6% |
7.4% |
NHS Trust data |
|
|
Total cost per event |
£17.28 |
£1,782 |
£3,507 |
|
|
Footnote: Expert opinion provided by panel of 5 anaesthetists, 5 obstetricians and 2 midwives.
Table 3: Clinical effectiveness estimates used in the economic model
|
Source |
PPH rate |
Use of additional uterotonic drugs |
||
|
|
Oxytocin |
Carbetocin |
Oxytocin |
Carbetocin |
|
Su 2012 (systematic review and meta-analysis) |
5.9% |
3.9% |
21.5% |
13.6% |
|
PPH defined as blood loss >500ml “or as defined by trialist”. Meta-analysis includes 4 studies; Borruto 2009 defines PPH >500ml, Boucher 1998 and Attilakos 2010 define PPH >1000ml, and Dansereau does not state definition. |
||||
Table 4: Overall cost as a result of using either oxytocin or carbetocin for prevention of post partumhaemorrhage at caesarean section
|
|
Oxytocin |
Carbetocin |
Change with Carbetocin |
|
PPH 500- 1000ml events |
65 |
43 |
-22 |
|
PPH 1000- 1500ml events |
13 |
9 |
-5 |
|
PPH >1500ml events |
10 |
6 |
-3 |
|
PPH Events |
88 |
58 |
-30 |
|
|
Oxytocin |
Carbetocin |
Change with carbetocin |
|
PPH Events |
£105,227 |
£69,242 |
-£35,985 |
|
PPH prophylaxis |
£3,600 |
£26,460 |
£22,860 |
|
PPH re-treatment |
£3,600 |
£2,250 |
-£1,350 |
|
Ante-Natal |
£124,312 |
£124,312 |
£0 |
|
Recovery |
£1,774,419 |
£1,761,637 |
-£12,783 |
|
Follow up |
£102,351 |
£102,090 |
-£261 |
|
Total cost |
£2,113,508 |
£2,085,990 |
-£27,518 |
